Small coloured organic compounds.
Water is not completely colourless, as we know, but let’s assume that it is, and that similar small organic compounds are colourless.
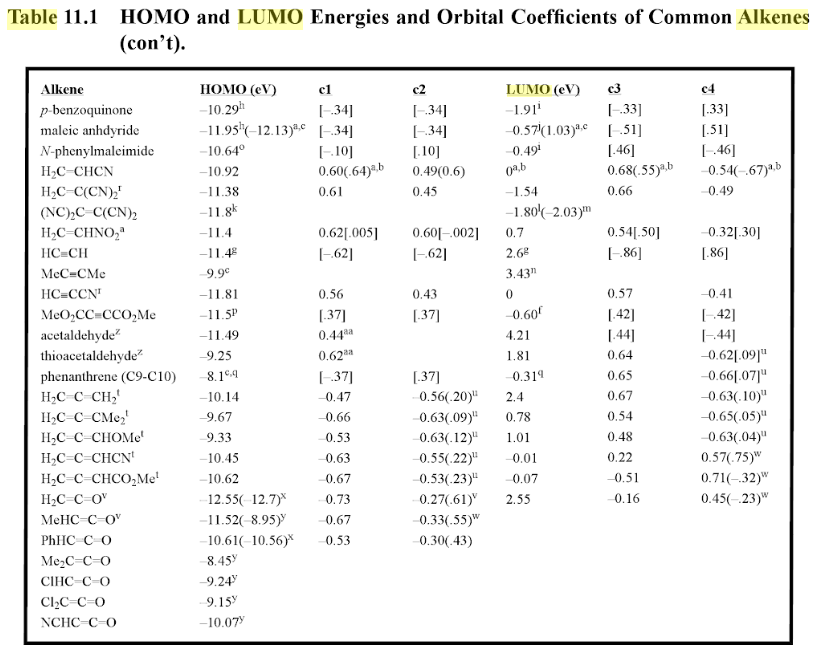
“The vast majority of simple organic compounds (e.g. ethanol) are colourless. Organic compounds tend to be coloured when there is extensive conjugation, causing the energy gap between the HOMO and LUMO to decrease, bringing the absorption band from the UV to the visible region. … HOMO and LUMO are types of molecular orbitals. The acronyms stand for highest occupied molecular orbital and lowest unoccupied molecular orbital. The energy difference between the HOMO and LUMO is termed the HOMO–LUMO gap”.
So, where can I find information about the size of the HOMO-LUMO gap for small coloured organic compounds?