We had a question on the SSSF FB page from John Perrier:
I have one litre of water ph 7 (neutral). I want to make it about ph 5 by adding vinegar of ph 2. How much should I add? TIA.
After a bit of discussion it emerged that he had been advised that one ml of vinegar per litre of water should do it (one part vinegar per thousand parts water, but that didn’t seem nearly enough vinegar to John.
My advice was that was not nearly dilute enough, and that it needed to be one part per million.
So check my working.
My reasoning was that pH is a negative power of ten, and that this meant there needed to be a 1000-fold reduction in H +. Because the acetic acid is a weak acid dividing into two ions, the equation of dissociation is going to mean you have to dilute by A 2 to divide the H + ion concentration by A.
Of course eventually you get to a point where the solution is so dilute that dissociation is complete, and it tends to act like an ordinary dilution process but my gut feeling was that’s not going to happen until the solution is weaker than pH = 5.
—-
Some explanation: CH3COOH is a weak acid, meaning that when you add it to water it doesn’t all break up into H + and CH3COO -. The extent of dissociation depends on the concentration: if the acetic acid solution is very concentrated, not much is dissociated, and when it is very dilute, it is almost all dissociated.
The equation is:
Ka = /
Ka is the acid dissociation constant which is 0.00001754 for acetic acid. Square brackets is molar concentration, ie moles per litre.
If y is the total molarity of acetic acid in solution and x is the molarity of hydrogen ions, then the amount of CH3COOH remaining will be y – x and the molarity of acetate ions will also be x, so you get
Ka = x^2/(y-x)
y – x = x^2/Ka
y = x^2/Ka + x
pH = -log(x)
For JP’s purpose, volume of acetic acid per volume of water is more useful than molarity so I worked that out using molar mass of acetic acid = 60.052 and density of acetic acid = 1049 g/L and assumed that volume was conserved on mixing which is not quite correct but the discrepancy is a third order effect.
So this is what that looks like with a log scale on the x-axis. The red line is a line where the goes as the square of the volume ratio, and the green line would be where is linear with the volume ratio. The curve is kind of parallel to the red line below a pH of maybe 3.7 and then it starts to peel off and by the time you get to a pH of 5.5. it’s similar to the green line. (As we get to pH higher than 6 this analysis won’t work so well because the autoionising potential of water becomes more important and the pH starts to curve down again: obviously, no matter how dilute the acetic acid, the pH is not going to go higher than 7. So I stopped around pH = 5.7).
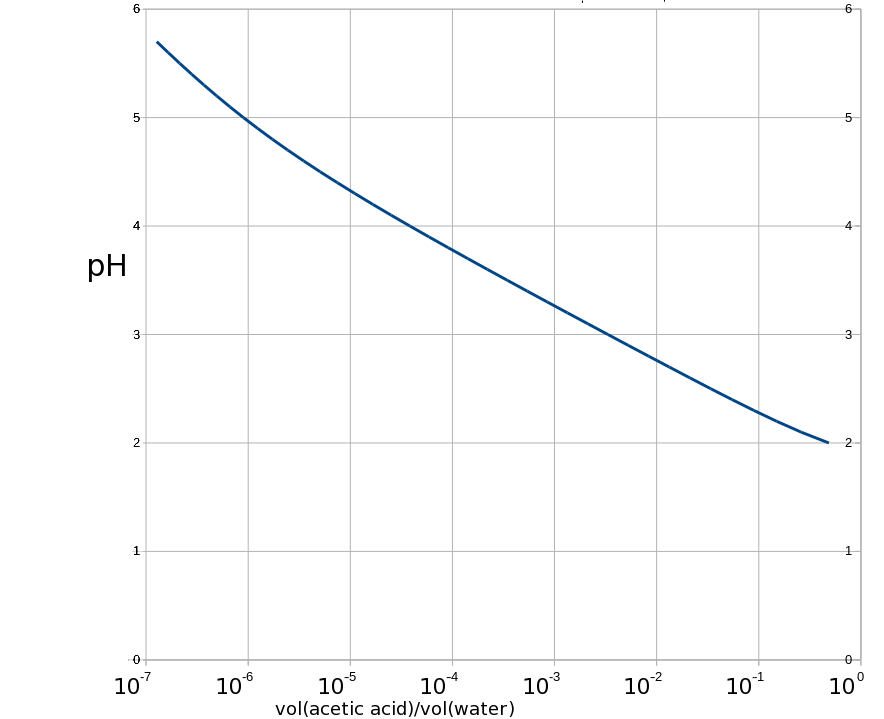
Anyway the dilution needs to be around 1 part of this pH = 2 vinegar per 540000 parts of water.
Now, ordinary household vinegar has a pH more like 2.5. I’d expect concentrated acetic acid as you might find in a lab’s reagent cabinet might be more like pH = 2. For this ordinary household vinegar the dilution is only 1 part per 38000.
Either way you’d probably need to use two steps to achieve this dilution. Like add a ml of vinegar to a litre of water, mix thoroughly, and THEN add some amount of THAT solution to a litre of water.